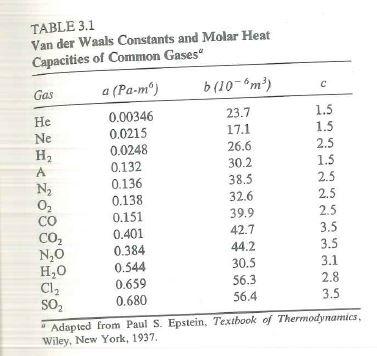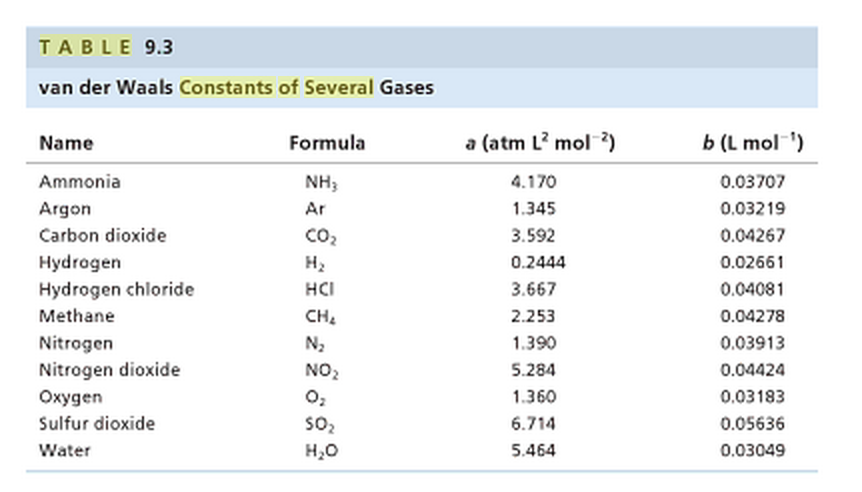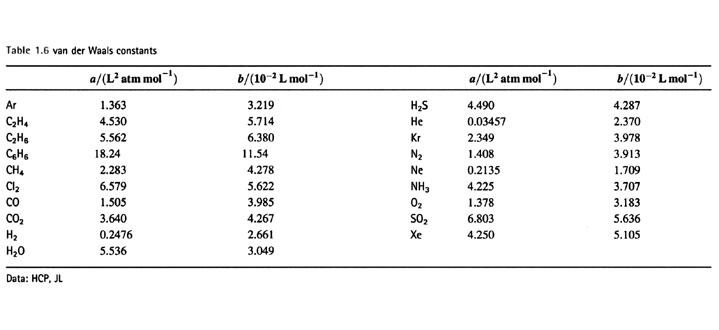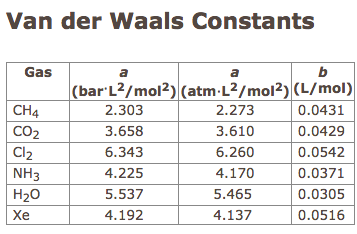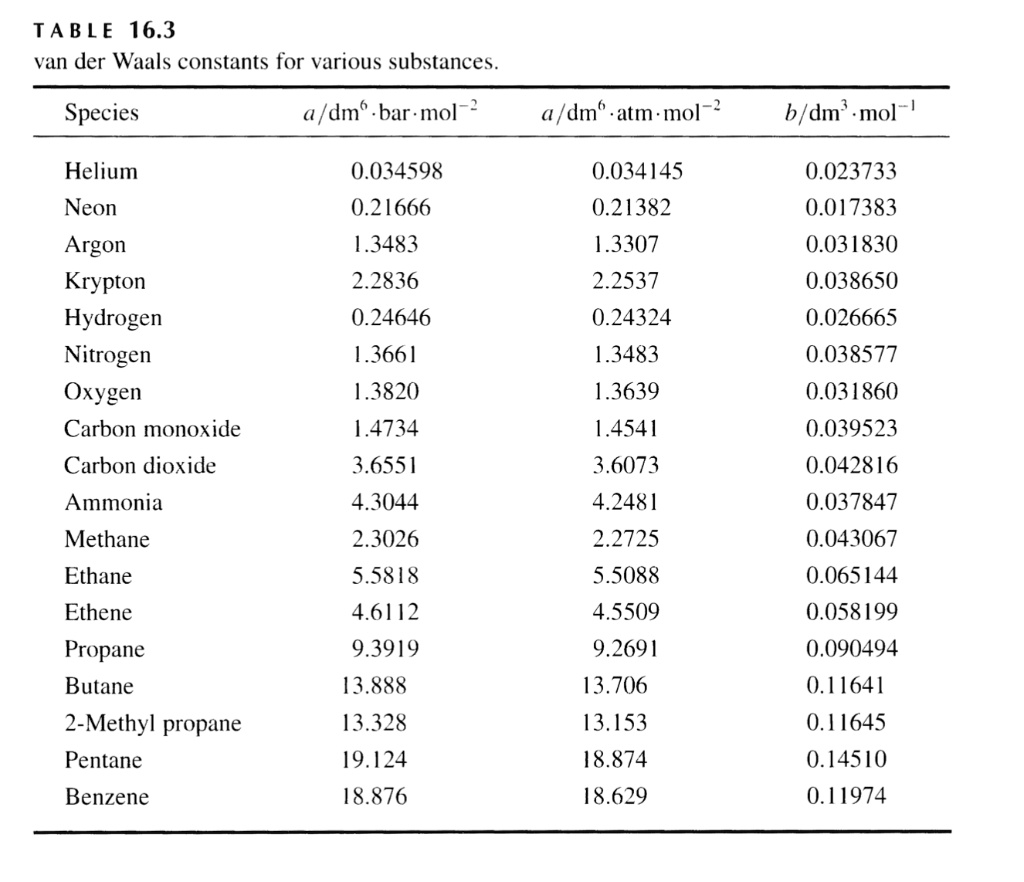
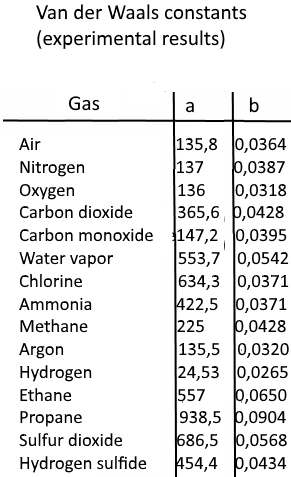
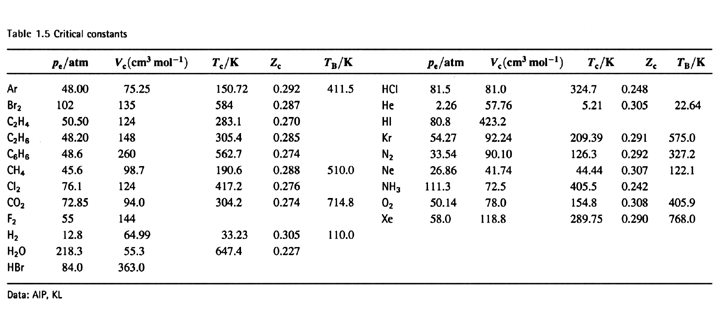
SOLVED: Table 16.3: van der Waals constants for various substances Species /dm^6 . bar-mol dm^3 . atm-mol b/dm^3 . mol Helium Neon Argon Krypton Hydrogen Nitrogen Oxygen Carbon monoxide Carbon dioxide Ammonia

SOLVED: The P, V, n, R, and T terms are the same as defined previously. The a term corrects for attractive forces between molecules while the b term corrects for the actual
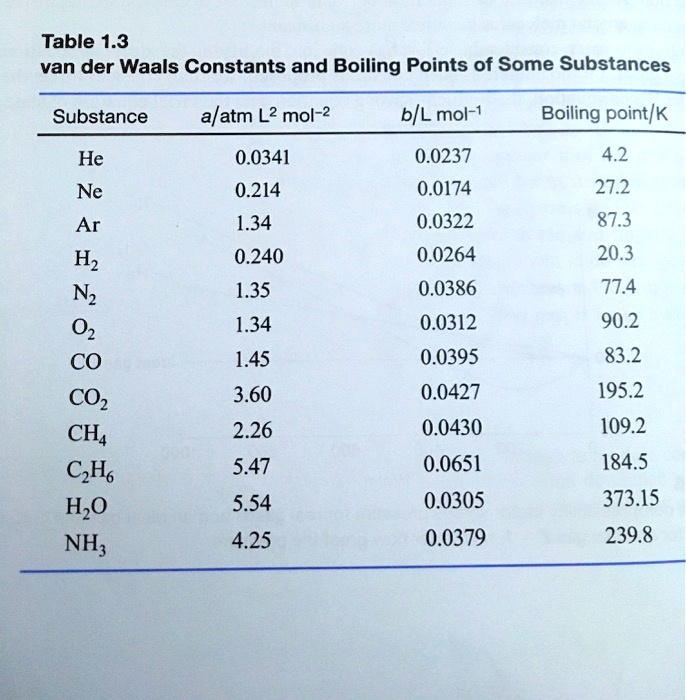
SOLVED: Table 1.3: van der Waals Constants and Boiling Points of Some Substances Substance alatm L? mol-2 b/L mol-1 Boiling point/k He 0.0341 0.214 1.34 0.240 1.35 1.34 1.45 3.60 2.26 5.47 5.54 4.25 0.0237 4.2 27.2 87.3 20.3 77.4 90.2 83.2 Ne 0.0174 ...
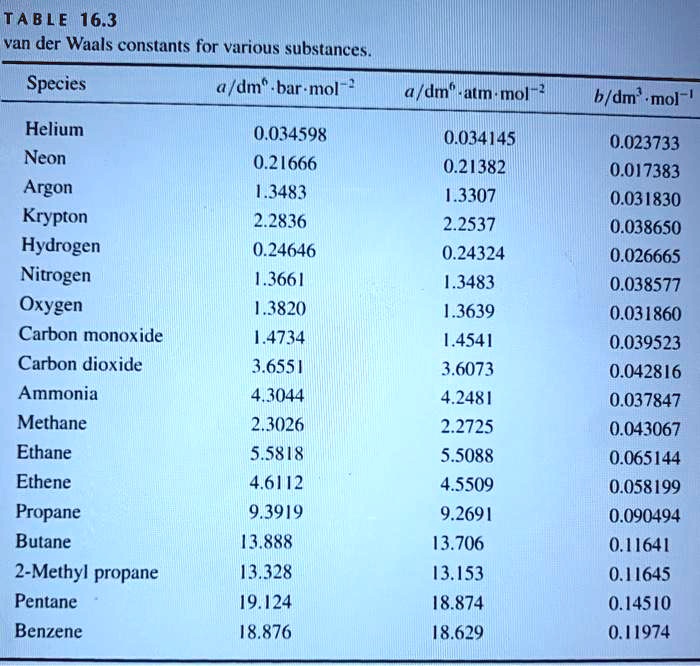

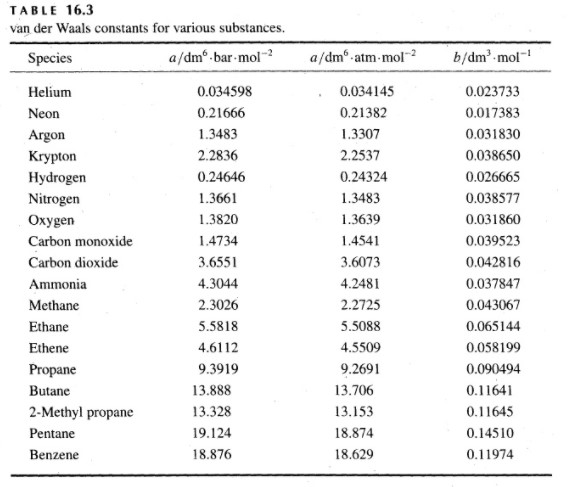




![Solved] Based on their respective van der Waals c | SolutionInn Solved] Based on their respective van der Waals c | SolutionInn](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/image/images11/876-(566).png)