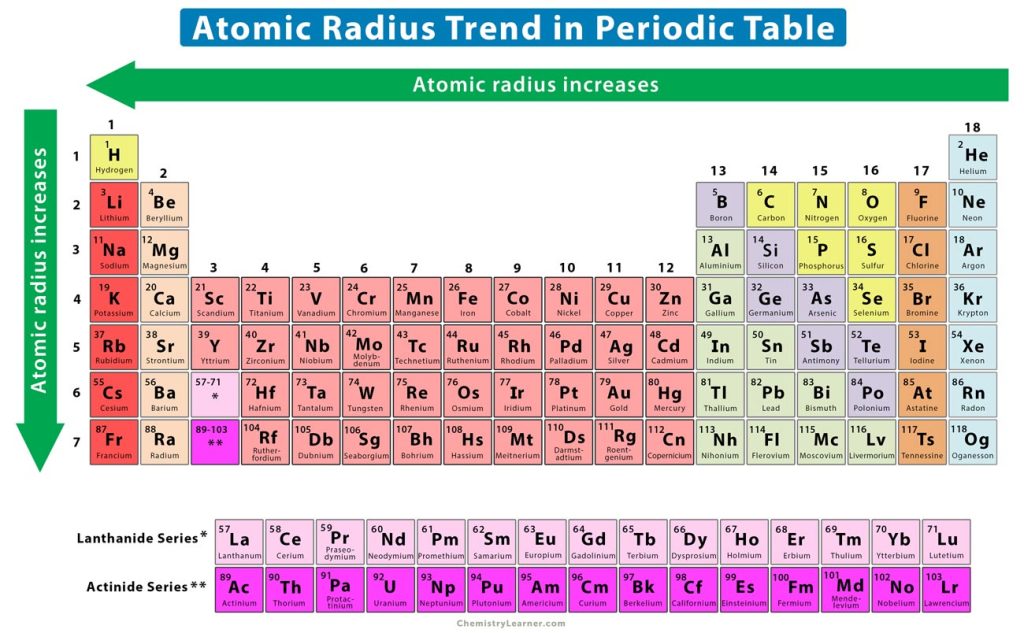
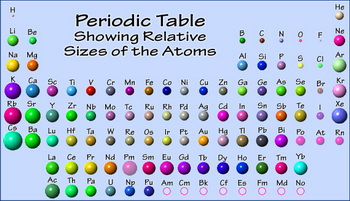
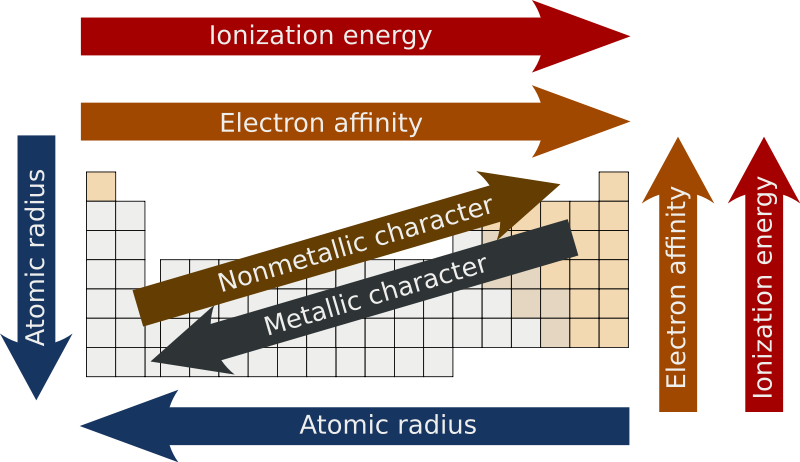
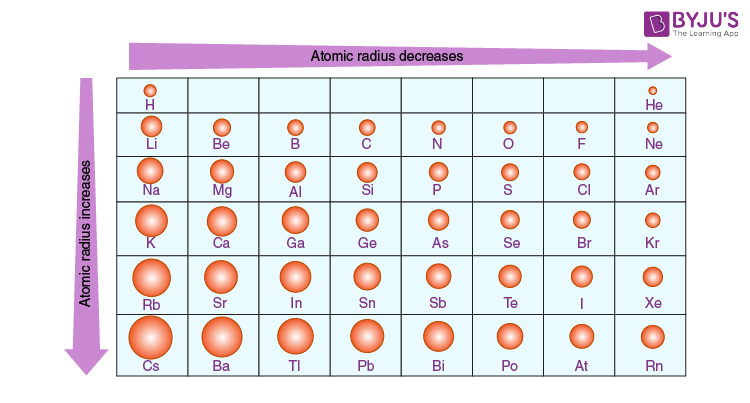
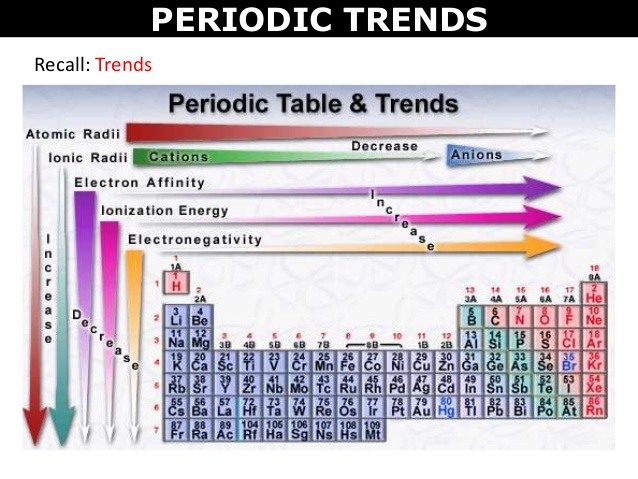
Trends in the Periodic Table (Chpt. 7). 1. Atomic radius (size) 2. Ionization energy 3. Electronegativity The three properties of elements whose changes. - ppt download

Which arrangement is in the correct order of radius size? a) Mn > Mn2+ > Cs b) Li+ > Li > Ra c) P < P3– < As3– d) Cr < Cr3+ < Ca e) Al3+ > Al > Si | Socratic

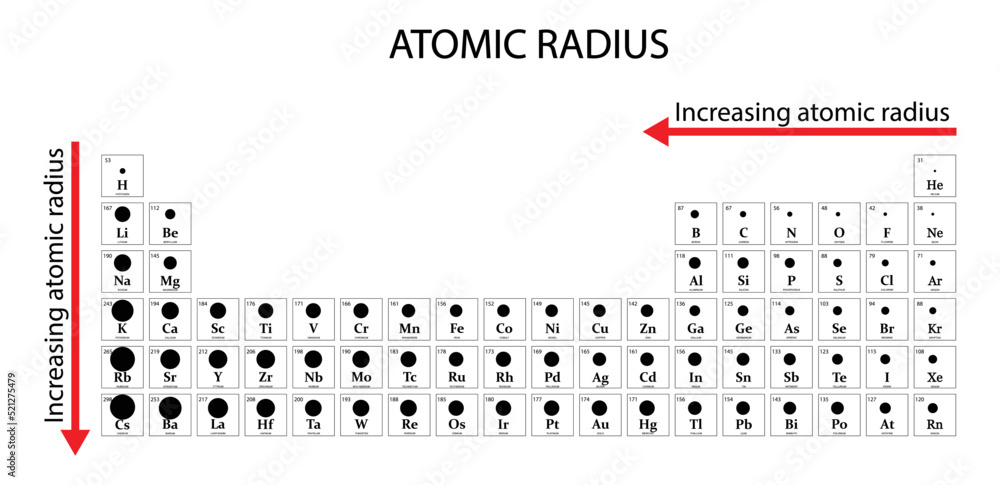
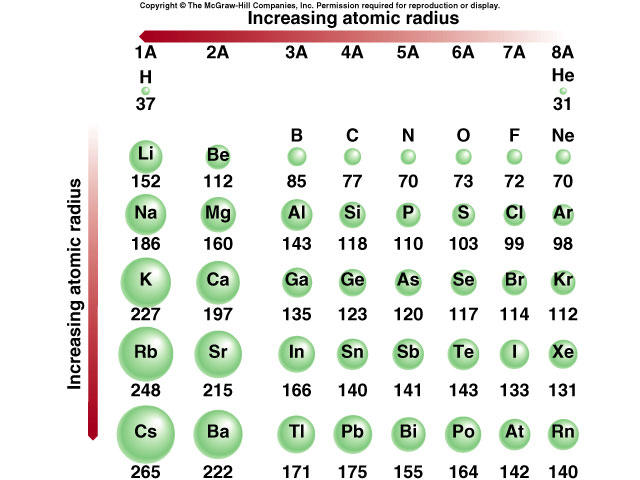
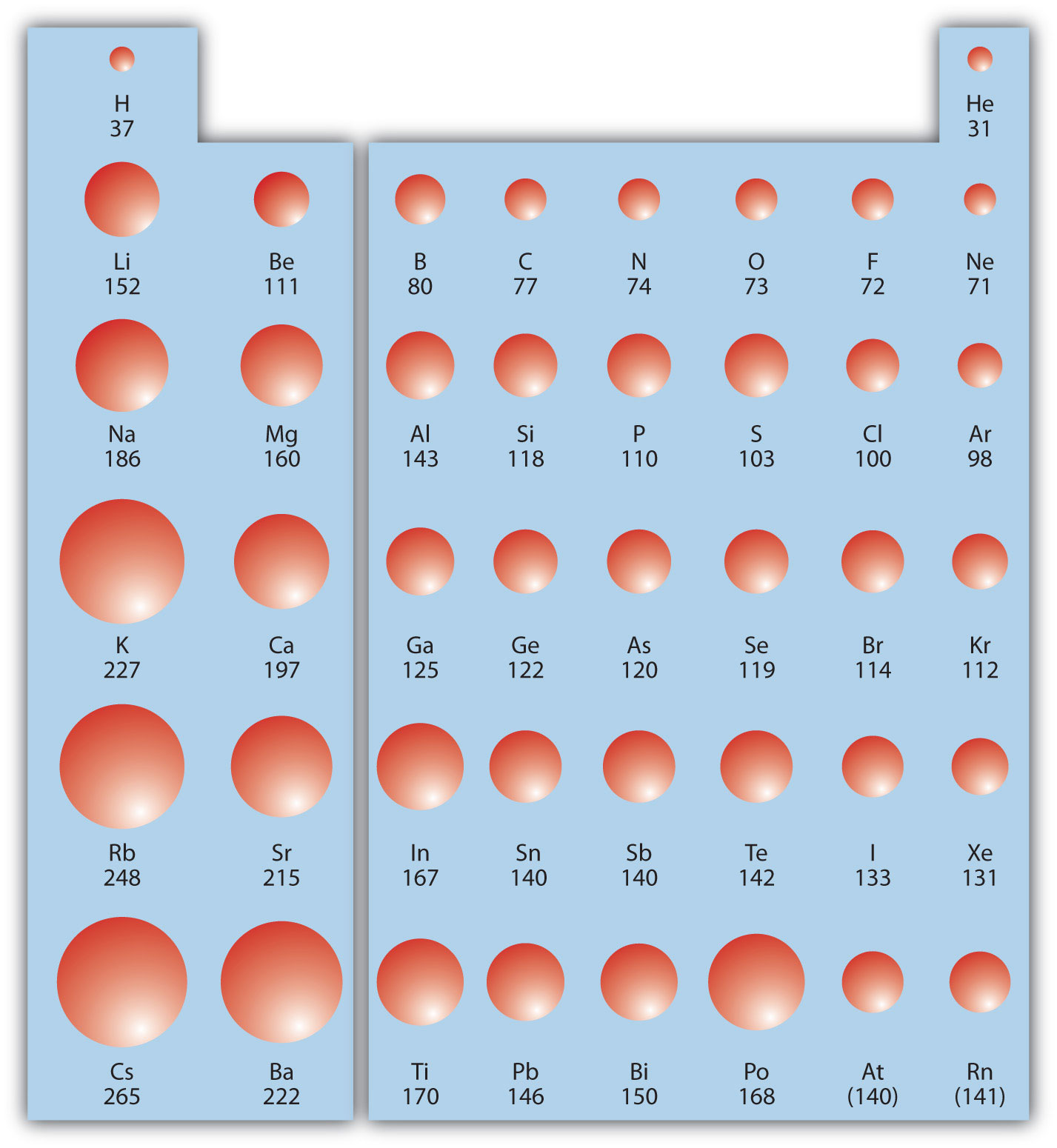
In the periodic table, Which element has the largest atomic radius? Which element has the smallest atomic radius? - 4zab3722




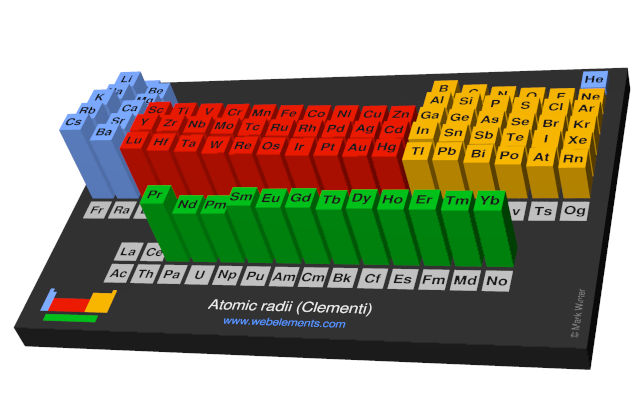


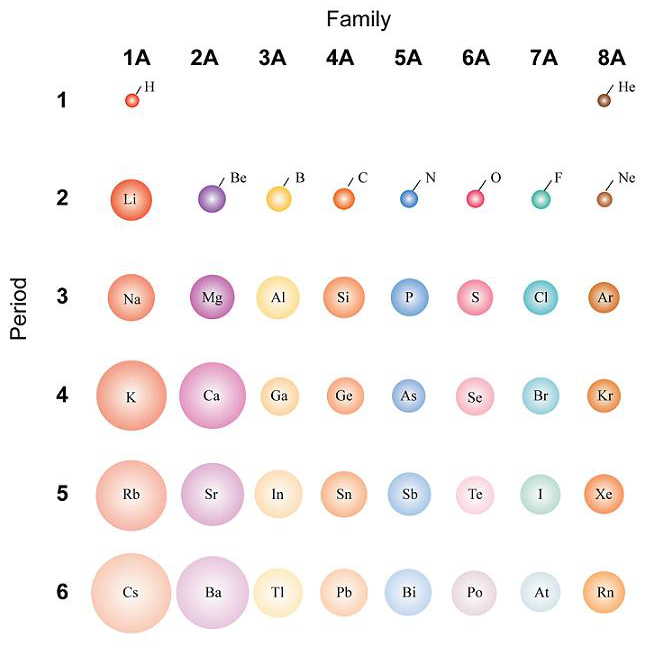
:max_bytes(150000):strip_icc()/chart-of-periodic-table-trends-608792-v1-6ee35b80170349e8ab67865a2fdfaceb.png)